Mosquitoes present one of India’s most prominent health threats because of the diseases they carry. Infectious diseases such as malaria, dengue fever, and recently, the Zika virus have caused much alarm in India and are responsible for several lakh deaths every year. Numerous novel methods have been suggested for curbing mosquito populations or reducing their capacity to spread disease. However, until now, nobody thought of putting them on a diet.
This bizarre approach was devised by Laura Duvall from Rockefeller University, when she decided to feed mosquitoes with experimental drugs designed to suppress the appetite of humans. “The whole thing started off as a joke,” says Leslie Vosshall, who led the subsequent study. “The assumption was that the human drugs would kill the animal or have no effect. It was a stupid thing”. It came as a surprise then, that the joke of an experiment seemed to work.
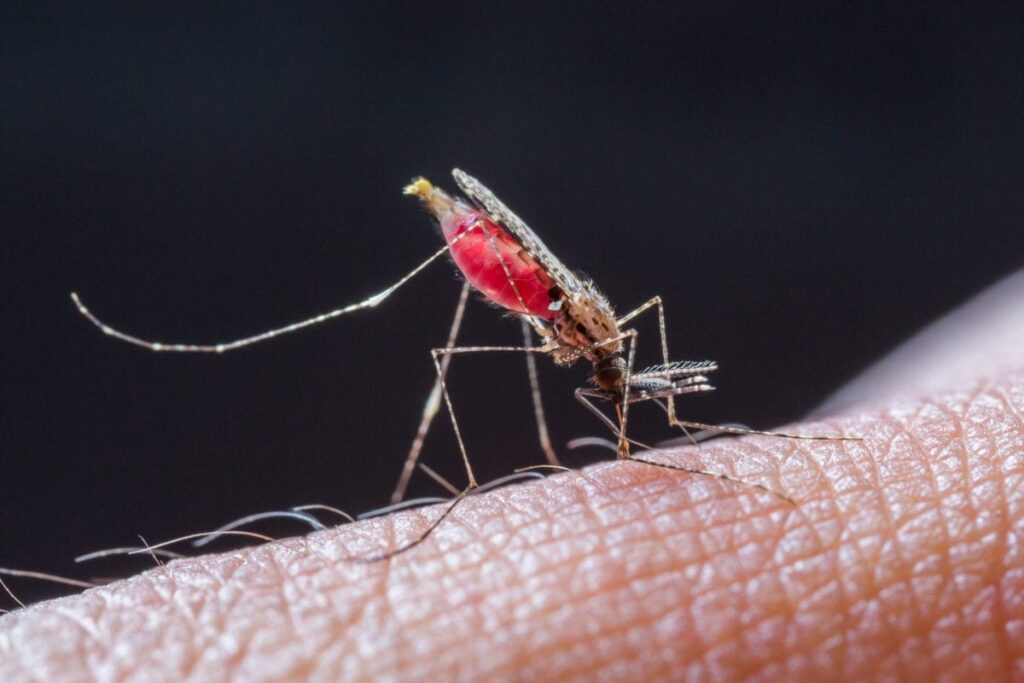
Mosquitoes and the spread of disease
The mosquitoes themselves are not the cause of these diseases. Rather, it is the numerous pathogens that utilise mosquitoes as a vector to transmit themselves that are the issue.
A mosquito population that is not infected with any pathogen may coexist with humans while causing no more issues to their neighbours than the nuisance of being bitten. However, the potential for an entire breeding population of mosquitoes to be infected with a pathogen — consequently passing on this pathogen to more people — makes every mosquito population a potential danger.
This is abundantly clear in densely packed Indian metropolises, where thousands are at risk of infection in just a small area. Transport links and the high number of individuals commuting across both the cities, and their immediate vicinity, can allow for a person infected with, for example, malaria, to be bitten again by a mosquito at any point along this root, and allow the infection to flourish in that area.
What if these mosquitoes stopped feeding?
Mosquitoes bite humans in order to feed; specifically the female of the species. The reason mosquitoes are so drawn to humans is the presence of a protein in human blood that is vital for the female mosquito to produce eggs. Once the mosquito has taken a blood meal from a human, they double their weight and enter a dormant state.
This dormant state is a vital part of the mosquito reproductive cycle. During this time the mosquito digests the blood, producing eggs in from the nutrients. Critically, at this stage of the mosquito reproductive cycle, they will have no instinct to continue to feed on humans. This stage that will last for days.
“Her interest in human cues is dialed down to zero,” Vosshall says. “You can put your hand in a cage of blood-fed females and you won’t get a bite.”
Vosshall began to wondering if this instinct that drives a mosquito either to feed or lay dormant could be altered via a chemical means. She focused on a small protein called neuropeptide Y, or NPY. This protein is found in a large number of species, with its role always being involved in appetite control. Its exact effect, however, varies among species: It drives flies and humans to eat but, as Duvall found, it does the opposite in mosquitoes, suppressing their appetite.
Among the pharmaceutical community the protein has been focused on as a means of producing diet controlling medication. As part of the study, the team procured drugs that target the same receptors as NPY, and began feeding them to mosquitoes in a saline solution.
Overall, the rate at which the mosquitoes fed on a human was reduced by around eighty percent following stimulation of the NPY receptor. By contrast, blocking the NPY receptor induced many mosquitoes who had already fed to attempt to continue to feed, despite already doubling their body weight in uptaken blood. This is a considerable finding that shows great promise in controlling mosquito populations.
Real world application
The negative connotation to this study is that its application to a real world breeding population of mosquitoes would likely kill off the mosquitoes, as their natural impulse to feed would be all but extinguished. This could have a negative impact on animals that feed on mosquitoes, as population size would be severely diminished.
However, controlled application may limit this negative effect. The most common animals to feed on mosquitoes are birds — feed on both the adult and larval forms — and fish, which feed on the larvae.
This ecosystem occurs to a greater degree in rural settings. In urban areas, it is far more likely that mosquitoes are forming breeding populations in stagnant water sources where there are no fish, such as artificial containers and sewage systems. This would mean that reduction of mosquito populations in urban areas would be far less impactful on the local ecosystem.
By highlighting mosquito populations in areas known for diseases such as malaria, this feeding prevention technique could be applied to limit transmission in a set area.
A real world example would be the recent Zika outbreaks in Rajasthan. Where Zika cases have been recorded, setting up mosquito traps in the area could limit further spread of the disease. “There are already surveillance traps that work by attracting mosquitoes,” says Vosshall. “We’d just need to engineer a feeding cup where we can put in our drug.”

